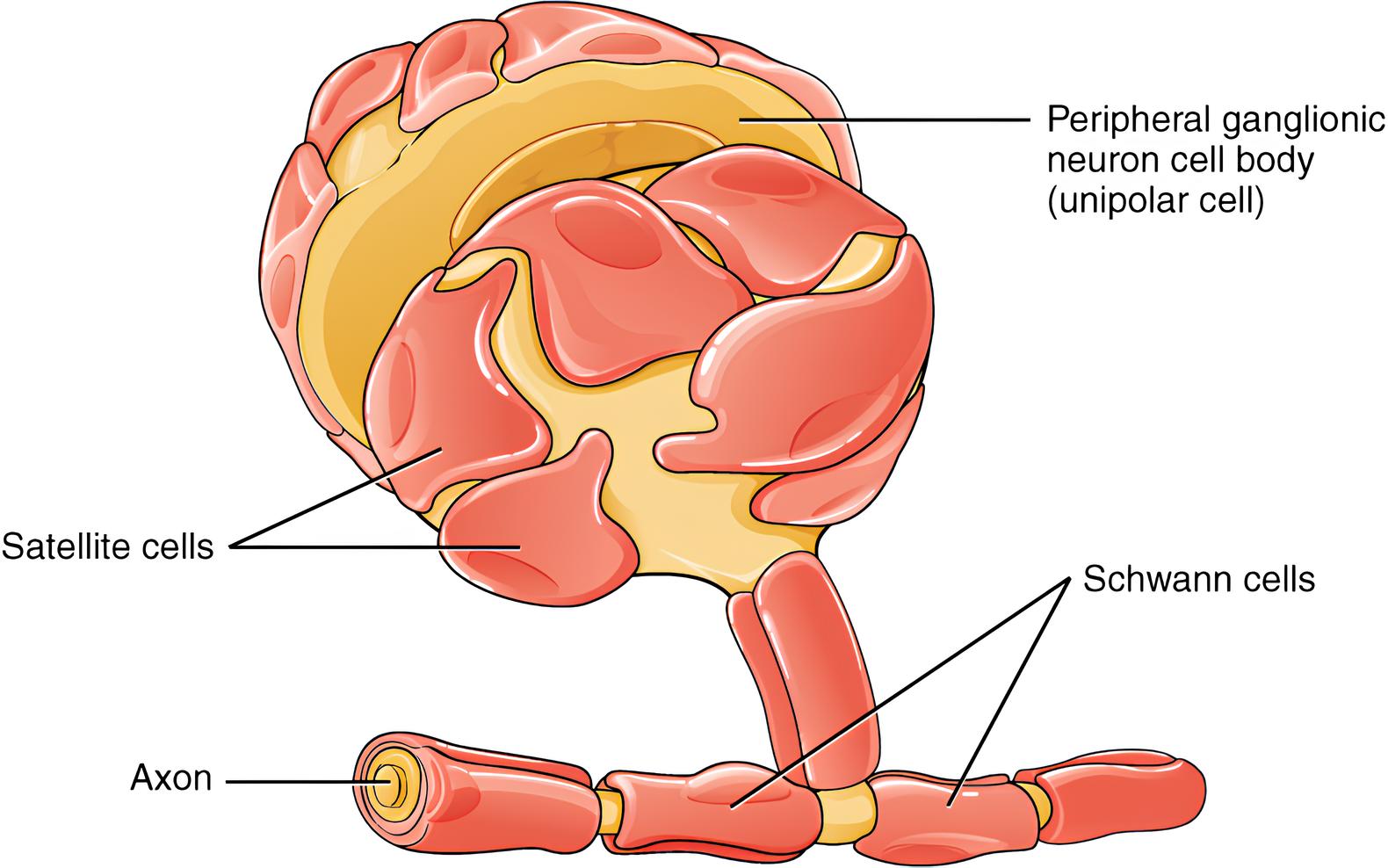
The network of peripheral nerves plays an essential role in transmitting signals and facilitating communication between different parts of the body. These nerves are responsible for conveying sensory information and enabling motor functions. However, injuries to these vital pathways can lead to significant disruptions in normal bodily functions. This is where the capabilities of Schwann cells come into play.
Schwann cells, which are a type of glial cell, have garnered attention for their pivotal role in aiding the repair and regeneration of damaged peripheral nerves. Unlike cells in other areas of the body, Schwann cells possess unique attributes that enable them to adapt and respond to nerve injuries. This inherent ability has earned them the title of “architects” of peripheral nerve repair.
In this article, we’ll explore the multifaceted role that Schwann cells play in the process of peripheral nerve repair. From facilitating the growth and regrowth of nerve fibers to creating an environment conducive to healing, Schwann cells are central players in the intricate mechanisms that promote nerve regeneration.
The Role of Schwann Cells in Nerve Regeneration
Schwann cells, situated within the peripheral nervous system, emerge as integral players in the intricate process of nerve regeneration. Their remarkable ability to adapt and respond to injuries contributes significantly to the restoration of nerve function.
Supporting the Formation of the “Band of Büngner”
When a peripheral nerve undergoes injury, Schwann cells immediately spring into action. They play a vital role in guiding the regenerating nerve fibers along the correct path. To accomplish this, Schwann cells line up in aligned rows, creating a supportive pathway known as the “Band of Büngner.” This organized arrangement acts as a conduit, helping to regenerate nerve fibers and bridge the gap formed due to injury.
Production of Growth Factors and Extracellular Matrix Components
Schwann cells go beyond mere structural support; they actively produce a variety of growth factors and extracellular matrix components. These growth factors serve as biochemical cues that stimulate nerve fiber growth and guide them along the appropriate trajectory. Furthermore, the extracellular matrix components provide a nurturing environment for regenerating nerve fibers, encouraging their development and integration into the existing neural network.
- Stimulation of Growth Factors
Schwann cells possess the capacity to generate growth factors, including nerve growth factor (NGF) and brain-derived neurotrophic factor (BDNF), among others. These factors act as signals that prompt neighboring nerve fibers to extend and elongate toward the target area. In this way, Schwann cells play a critical role in coordinating the directional growth of regenerating nerves.
- Provision of Extracellular Matrix Support
The extracellular matrix, which Schwann cells secrete, functions as a scaffold to support regenerating nerve fibers. This matrix provides physical and chemical guidance, aiding in the alignment and orientation of growing nerve fibers. By offering a conducive environment enriched with essential molecules, Schwann cells actively participate in the process of neural reconstruction.
Schwann Cells’ Adaptive Abilities
Schwann cells, situated within the peripheral nervous system, showcase a remarkable capacity for adaptation in response to nerve injuries. These adaptive abilities enable them to actively contribute to the process of nerve repair and regeneration.
Dedifferentiation and Migration to Injury Sites
Upon encountering a nerve injury, Schwann cells exhibit a unique capability called dedifferentiation. In simple terms, they revert to an earlier developmental state that enables them to respond effectively to the injury. This dedifferentiation process prompts Schwann cells to undergo morphological changes, facilitating their migration toward the site of injury. This migration is a crucial step in preparing the cellular environment for subsequent regenerative processes.
Creation of a Supportive Microenvironment
Schwann cells, as adaptable entities, don’t merely migrate to the injury site; they also engage in creating a supportive microenvironment for nerve regeneration. They actively release various molecules that enhance the local environment’s suitability for regrowth. By modifying the extracellular conditions, Schwann cells encourage the survival and growth of regenerating nerve fibers, contributing to successful repair.
Role in Axon Guidance and Synapse Formation
In addition to their role in supporting the physical structure of regenerating nerves, Schwann cells also participate in axon guidance and synapse formation. As nerve fibers extend and navigate toward their target areas, Schwann cells provide guidance cues that assist in proper alignment. This guidance ensures that the regenerated nerves accurately connect to their intended destinations, restoring functional pathways.
Myelination and Remuscularization
Schwann cells, integral to the peripheral nervous system, play a role not only in nerve repair but also in processes like myelination and remuscularization. These functions contribute to the proper functioning and restoration of damaged nerves.
Myelination of Nerve Fibers
One of the pivotal roles of Schwann cells is to facilitate the myelination of nerve fibers. Myelin, a protective and insulating sheath, is essential for efficient nerve signal transmission. Schwann cells wrap themselves around nerve fibers, creating compact layers of myelin that act as insulation. This myelin sheath enhances the speed and efficiency of nerve impulses, enabling rapid communication between nerve cells. Schwann cells not only myelinate single nerve fibers but also guide the process of myelin sheath formation, ensuring precision and functional integrity.
Contribution to Remyelination After Injury
In cases of nerve injury that result in myelin damage or loss, Schwann cells once again demonstrate their adaptability. They actively participate in the process of remyelination, which involves the regeneration of the myelin sheath. Schwann cells mobilize to the injured site and initiate the formation of new myelin layers around the regenerating nerve fibers. This process aims to restore optimal signal conduction, allowing for the reestablishment of efficient communication between nerve cells.
Factors Influencing Schwann Cell-Mediated Regeneration
The process of Schwann cell-mediated nerve regeneration is influenced by a range of factors that contribute to the success and efficiency of this process. Understanding these factors provides insights into the complexity of nerve repair and the role Schwann cells play.
Impact of Age, Location, and Type of Nerve Injury
- Age: Age is a determinant in the regenerative potential of Schwann cells. Younger individuals tend to have more robust regenerative capabilities compared to older individuals. Schwann cells in younger bodies exhibit greater responsiveness to injury signals and are more effective in promoting nerve repair.
- Location of Injury: The location of the nerve injury within the body plays a significant role in Schwann cell-mediated regeneration. Nerves in certain regions, such as the limbs, may have different regenerative capacities compared to nerves in the central nervous system. The microenvironment and surrounding tissues also influence the effectiveness of Schwann cell activity.
Interaction with Other Cells
- Macrophages: Schwann cells interact with immune cells, particularly macrophages, during the regenerative process. Macrophages assist in clearing debris and providing an environment conducive to repair. The interplay between Schwann cells and macrophages is essential for orchestrating an effective regenerative response.
- Neighboring Nerve Cells: The presence of neighboring nerve cells also influences Schwann cell-mediated regeneration. Communication between nerve cells and Schwann cells is vital for coordinating regrowth and ensuring accurate reconnection. Signals from neighboring nerve cells guide Schwann cells in the direction of proper nerve regeneration.
- Cytokines and Growth Factors: Various signaling molecules, including cytokines and growth factors, modulate the behavior of Schwann cells. These molecules regulate Schwann cell activity and influence processes like migration, proliferation, and the secretion of factors that aid in nerve repair.
Current Research and Clinical Applications
The field of Schwann cell research continues to evolve, with ongoing studies exploring innovative ways to harness their regenerative potential for clinical applications. Advances in understanding Schwann cell behavior have led to promising developments in the realm of nerve repair and therapeutic interventions.
Ongoing Studies in Schwann Cell-Based Therapies
- Tissue Engineering Approaches: Researchers are investigating tissue engineering strategies that involve utilizing Schwann cells to create artificial nerve conduits. These conduits can serve as guides for regenerating nerve fibers, providing a scaffold along which new nerves can grow. This approach holds promise for treating nerve injuries and promoting functional recovery.
- Gene and Cell Therapies: Gene and cell therapy approaches are being explored to enhance Schwann cell functionality. By introducing specific genes or manipulating cellular behavior, researchers aim to optimize the regenerative potential of Schwann cells. This can lead to improved nerve repair outcomes and accelerated healing.
Potential Applications in Nerve Injuries and Disorders
- Peripheral Nerve Injuries: Schwann cell-based therapies have shown promise in the treatment of peripheral nerve injuries. By harnessing Schwann cells’ ability to create a conducive environment for nerve regeneration, these therapies offer hope for individuals recovering from nerve trauma.
- Neuropathies and Neurodegenerative Disorders: Researchers are investigating the potential of Schwann cell-based interventions in addressing neuropathies and neurodegenerative disorders. These cells could potentially play a role in promoting nerve cell survival, restoring neural connections, and alleviating symptoms associated with these conditions.
- Spinal Cord Injuries: While challenging, Schwann cell transplantation is being explored as a potential avenue for spinal cord injury treatment. The regenerative properties of Schwann cells may contribute to remyelination and axon regrowth, ultimately aiding in restoring functionality after spinal cord damage.
Conclusion
In the realm of nerve repair and regeneration, Schwann cells emerge as pivotal players, driving the process of healing and restoration. Their remarkable adaptability, along with their multifaceted roles in myelination, remuscularization, and nerve guidance, underscores their significance in maintaining neural connectivity. As we conclude our exploration of Schwann cells’ contributions, let’s reflect on the key takeaways and implications:
A Dynamic Force in Nerve Regeneration
Schwann cells exhibit a dynamic response to nerve injuries, showcasing their capacity to adapt and create an environment conducive to repair. Their ability to dedifferentiate, migrate to injury sites, and release growth factors underscores their role as architects of the regenerative process. By actively supporting the formation of the “Band of Büngner” and orchestrating the production of growth factors, Schwann cells contribute to the coordinated growth of regenerating nerve fibers.
Shaping the Landscape of Clinical Applications
The ongoing advancements in Schwann cell research hold significant promise for translating laboratory findings into real-world clinical applications. From tissue engineering approaches to gene and cell therapies, researchers are working diligently to harness Schwann cells’ potential for treating nerve injuries, neuropathies, and even neurodegenerative disorders.
Schwann Cells at a Glance
What is the myelin sheath, and how do Schwann cells contribute to its formation?
How do Schwann cells contribute to nerve regeneration?
Are Schwann cells the only cells responsible for myelination?
Can Schwann cells also perform other functions besides myelination?
How do Schwann cells interact with axons?
Are there any disorders associated with Schwann cells?
References
- Zhang, X., Jiang, X., Jiang, S., Cai, X., Yu, S., & Pei, G. (2021, July 7). Schwann cells promote prevascularization and osteogenesis of tissue-engineered bone via bone marrow mesenchymal stem cell-derived endothelial cells – Stem Cell Research & Therapy. BioMed Central. https://doi.org/10.1186/s13287-021-02433-3
- Huang, Z., Powell, R., Phillips, J. B., & Haastert-Talini, K. (2020). Perspective on Schwann Cells Derived from Induced Pluripotent Stem Cells in Peripheral Nerve Tissue Engineering. Cells, 9(11), 2497. https://doi.org/10.3390/cells9112497
- Featured Image: Version 8.25 from the Textbook
OpenStax Anatomy and Physiology.